
Prevalence of Serum Antibody Titers against Core Vaccine Antigens in Italian Cats
Prof. Paola Dall’Ara DVM, PhD
Associate Professor of Veterinary Immunology and Infectious Diseases of Dogs and Cats
Department of Veterinary Medicine and Animal Sciences, University of Milan, Italy
General Introduction:
Feline Panleukopenia (FPV), Herpesvirus type 1 (FeHV-1), and Calicivirus (FCV) are highly infective and significant health risks resulting in high rates of morbidity and mortality in cats worldwide. Vaccination is considered the best method for preventing infection with these diseases.
Summary
Background
Veterinary medical associations worldwide have created guidelines that establish core vaccination as the most significant and effective method for preventing widespread, infectious diseases. Vaccines developed for FPV, FeHV-1, and FCV are the three recommended feline core vaccines. For decades, feline core vaccines have been administered on an annual basis; however, with the advent of new formulations, medical professionals now suggest less frequent vaccinations, with either modified live (attenuated) core vaccines (MLV) or killed core vaccines that are licensed for triennial use.
Since the duration of immunity appears to decrease with time and vaccine efficiency varies by individual, it appears reasonable to vaccinate cats using a tailored approach based on their lifestyle. High-risk cats (indoor/outdoor, multi-cat family) would be vaccinated more frequently (every 1-2 years) under this protocol, while low-risk cats (solitary indoor) would be vaccinated every 3 years.
A multitude of factors impede the development of a sufficient immune defense. It is possible to reduce the incidence of vaccination failures and unnecessary vaccinations by being aware of the kitten, adult, and elderly cat’s immune status. Knowing the true immune status of each cat with regard to core vaccines could assist veterinarians in selecting the best vaccination protocol for each feline patient.
The use of in-clinic tests for the purpose of core vaccine titer testing in cats is still very limited with only a few peer-reviewed publications in professional journals.
Aim
The aim of this study was to determine the actual protection against FPV, FeHV-1, and FCV in a large cohort of Italian cats by using the in-practice titer test kit Feline VacciCheck.
Methods
A total of 740 feline serum/plasma samples from owned cats (567) and stray cats (173) were included in the study. The ages ranged from 4 months to 25 years with 14.7% kittens, 47.6% young adults, 15.1% mature adults, and 22.6% seniors. Samples were tested for the presence of Protective Antibody Titers (PATs) for FPV, FeHV-1, and FCV using the in-clinic titer test Feline VacciCheck.
Interpretation of Test Results
Antibody concentrations were defined as “S” units (S1-S6) which are related to the color intensity of the resulting spots produced with this dot-ELISA titer test. The color intensity of each test spot corresponds directly to the antibody level in the sample tested. The S values have been standardized by the manufacturer for all 3 viruses. Results were divided into four categories (unprotected, weakly positive, medium positive, and high positive) based on the threshold values of each pathogen.
Results
The percentage and the number of cats belonging to the different categories of protection against FPV, FeHV-1, and FCV of the whole population appears in Graph 1. Graph 2. demonstrates the immune status of the two categories (origin) of cats tested.
Results
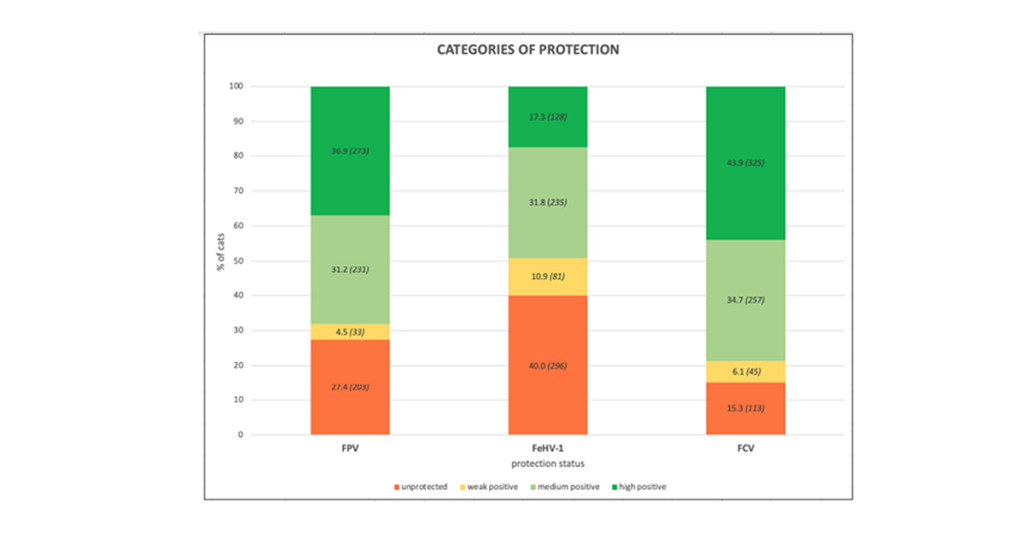
Graph 1. The % and number of cats according to categories of protection against FPV, FeHV-1, and FCV.
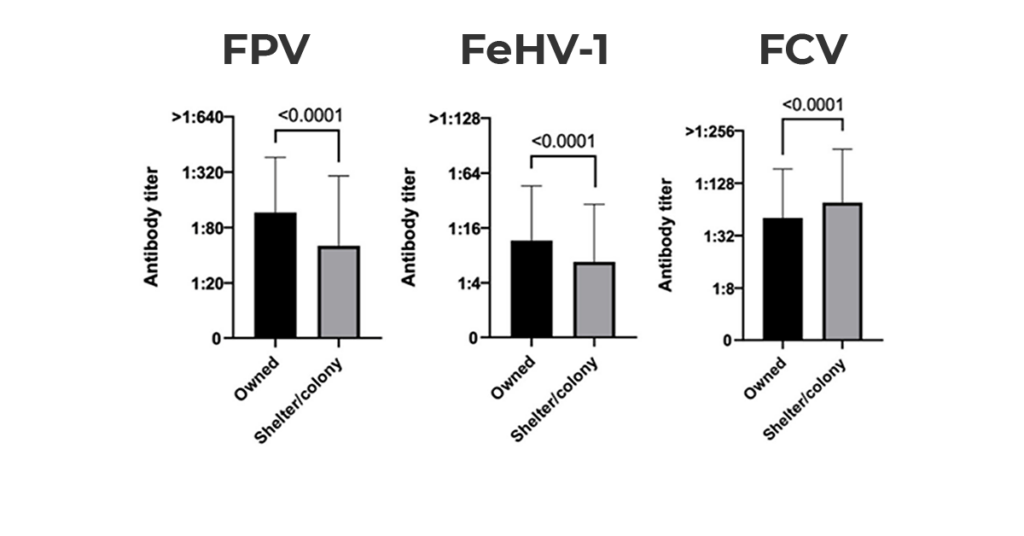
Graph 2. Antibody titers against FPV, FeLV-1 and FCV according to origin
Conclusions
Many feline core vaccines are registered with a 3-year duration of immunity which was derived from post-challenge protection studies. The benefits of vaccination should always outweigh the risk of adverse reactions such as Feline Injection–Site Sarcoma. The decision to vaccinate and boost with core vaccines should be based on a careful assessment of the likelihood of exposure, the severity of disease, as well as the risk/benefit ratio for each disease and the individual cat.
When possible, an assessment of specific antibody titers should precede the decision to apply a shorter time span between vaccination (1 to 2 years) in cats at high risk. In most cases, the use of antibody titration to estimate protection before or after a core vaccination should be the first choice of every veterinary practitioner in daily practice.
The feline in-clinic test used in this study (Feline VacciCheck) provided reliable and trustworthy results and confirms itself as a valid tool in everyday veterinary practice.


